
Management of acquired Haemophilia A: A case series
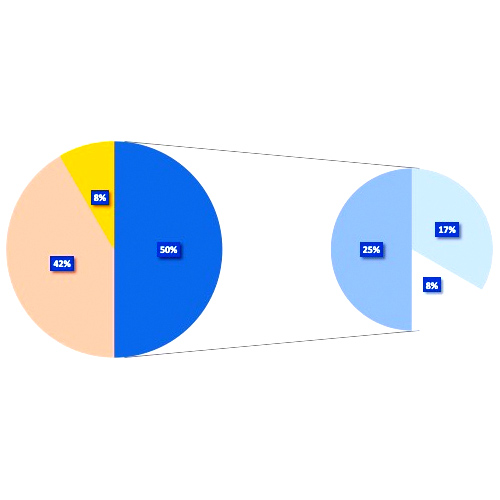
ACQUIRED HAEMOPHILIA A: MAIN FEATURES Acquired haemophilia A (AHA) is a rare hematologic disorder potentially leading to life-threatening bleeding. It is caused by inhibitors against factor VIII (FVIII). AHA typically manifests itself through spontaneous or...
Immune Tolerance Induction with Nuwiq® (Simoctocog Alfa) in Nine Patients with Severe Haemophilia A and Inhibitors to FVIII
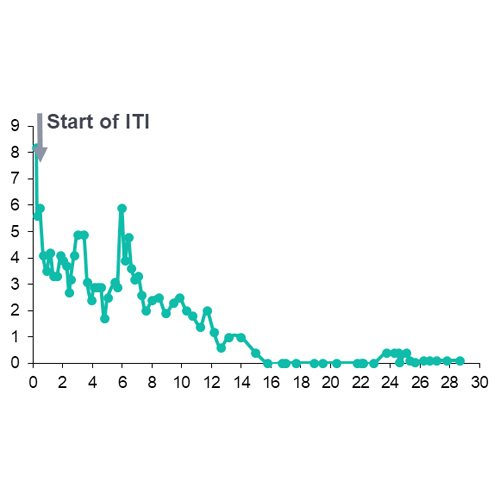
Introduction The risk of developing inhibitors to coagulation factor VIII (FVIII) remains a serious concern when treating children with haemophilia A Immune tolerance induction (ITI) with FVIII is the only proven approach to eradicate inhibitors1,2 Human cell...
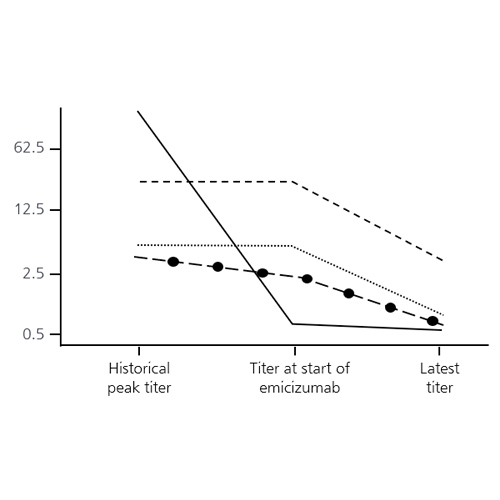
Combination of emicizumab with simoctocog alfa for prophylaxis in previously untreated/minimally treated hemophilia A patients, and for managing inhibitor patients
The combination of plasma-derived FVIII/VWF with emicizumab has non-additive effects on thrombin generation assay independently of the presence of inhibitors in hemophilia A plasma
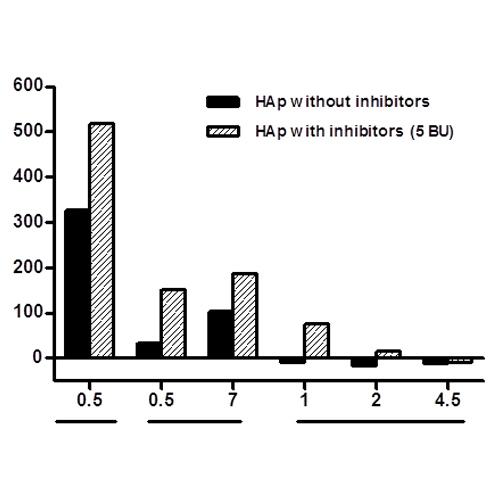
BACKGROUND Emicizumab (Hemlibra®, Chugai-Roche) is used to bridge aFIX and FX to function as missing aFVIII in hemophilia A (HA) patients. Although limited clinical results are available, fatal thrombotic complications have been reported when using emicizumab combined...
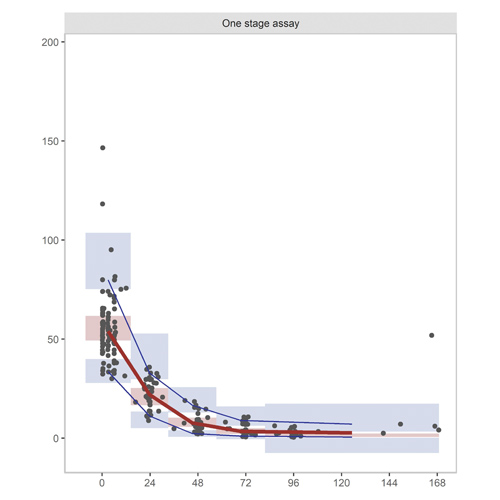
Population pharmacokinetic model for recombinant factor VIII Fc fusion protein (rFVIIIFc) validated and optimized for use in children
Background Pharmacokinetic(PK)-guided dosing can aid in individualizing replacement therapy for hemophilia patients. The current population PK models used for PK guided treatment are based on clinical trial data, whereas real clinical patients may differ significantly...
Final results from the NuProtect study of Nuwiq® (simoctocog alfa) treatment in previously untreated patients with severe haemophilia A
Introduction Neutralising antibodies against exogenous clotting factor VIII (FVIII) remain the most significant treatment complication in patients with haemophilia A, particularly in previously untreated patients (PUPs)1-4 Inhibitors develop in approximately 35% of...

Prognostic value of polymorphisms of the CYP2C19 gene in the development of stent thrombosis
Background The consequence of insufficient suppression of increased activity of platelets can be a repeated cardiovascular event. At the heart of the variability of the pharmacological response of clopidogrel are many factors, among which the most important is the...
Overview of efficacy and safety of a plasma-derived human von Willebrand factor concentrate for perioperative management and delivery in patients with hereditary VWF deficiency, unresponsive to dDAVP
INTRODUCTION WILFACTIN®/WILLFACT® is a plasma-derived von Willebrand factor (pdVWF) concentrate almost devoid of factor VIII (FVIII)*. The concept of a FVIII-poor VWF concentrate is based on the principle that what is missing in VWD patients is VWF only and not FVIII....
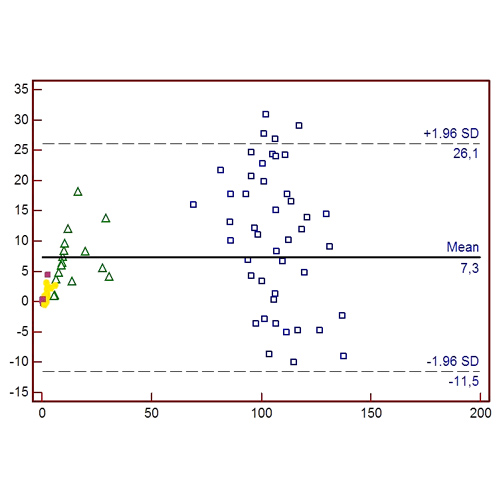
Evaluation of the chromogenic assay “Biophen factor IX” on Sysmex CS-2400 analyzer
BACKGROUND and AIM Measurement of Factor IX is required for quantification of residual plasma FIX levels with a diagnostic purpose (Haemophilia B) or for performing recovery studies during treatment by replacement therapies. The one-stage clotting assay is the most...
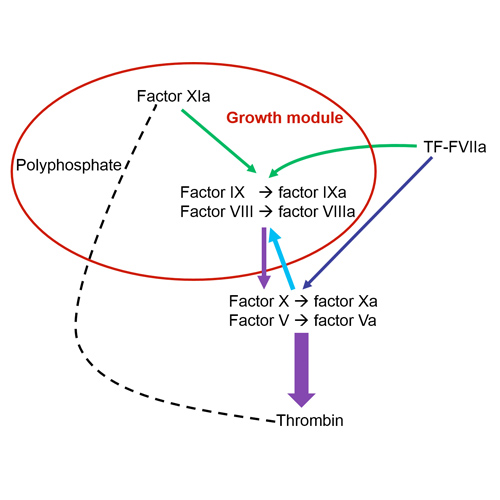
Testing clot growth and thrombin wave patterns in haemophilia patients
Introduction Thrombodynamics is a new pharmacodynamic test system for coagulation. Clot growth and thrombin wave patterns in space are documented with a video-microscopic system with a fluorogenic thrombin substrate. Coagulation is activated by immobilized tissue...
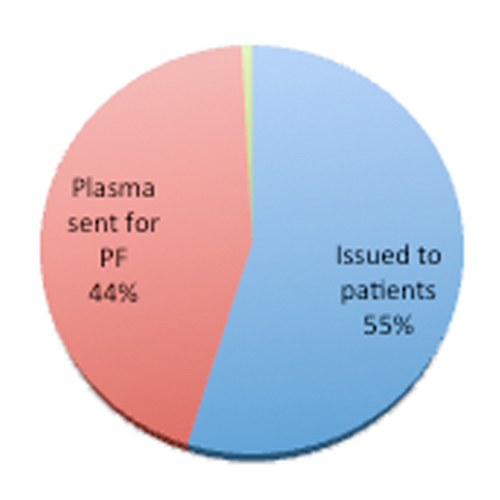
Six years of experience with plasma fractionation industries at a private super speciality hospital in South India: renaissance in usage of excess plasma in blood banks
Background According to World Health Organization (WHO) each country must plan for its safe and consistent supply of blood and plasma products for regular clinical needs as well as in case of any disaster. Self-sufficiency ratio is the key parameter to assess the...